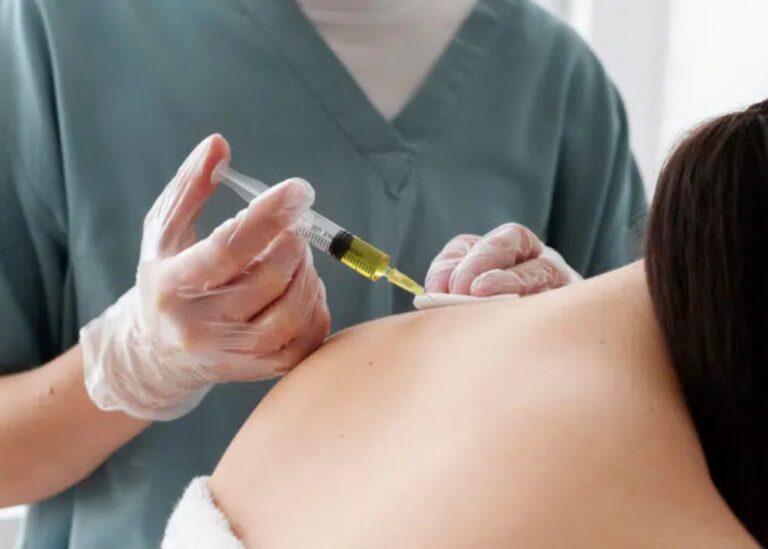
When considering stem cell therapy, the first question is always: Is it safe? The answer depends entirely on how it’s performed.
In proper medical settings with qualified doctors using screened cells, stem cell therapy has a strong safety record. Clinical research demonstrates that serious adverse events like death or infection are not significantly higher in stem cell recipients compared to control groups. Outside legitimate oversight, however, the risks become serious, and unqualified clinics present documented safety hazards.
What Determines Safety
Safe versus unsafe stem cell therapy comes down to three things: cell quality, where therapy happens, and who performs it.
Unqualified clinics cut corners by using cells from unknown sources, skipping safety screening, or performing procedures outside medical environments. Many unlicensed clinics operate without proper oversight, creating significant risks. Legitimate clinics operate under government oversight: the FDA in the U.S., COFEPRIS in Mexico.
What Safe Stem Cell Therapy Includes
Safe stem cell therapy starts with cell quality and sourcing. At RMRM, we use autologous stem cells harvested from your own adipose (fat) tissue, a rich source of regenerative cells that eliminates rejection risks. Your cells are concentrated using minimally manipulated processing and deployed precisely where needed using image guidance.
Before reaching patients, cells undergo rigorous screening:
- Donors are screened for infectious diseases
- Cells are cultivated in FDA-certified, sterile laboratories
- Final products are tested for contamination and viability
- Every batch includes a Certificate of Analysis (COA) documenting purity, potency, and viability testing
Legitimate procedures happen in licensed medical facilities with strict sterile protocols, performed by licensed medical doctors who evaluate your specific health situation and design personalized plans.
Read More: Stem Cell Therapy Side Effects
Side Effects and Safety Profile
When stem cell therapy is performed properly with quality cells, side effects are almost always mild and short-lived.
Common, Mild Reactions: Most frequent side effects include mild injection site reactions, temporary low-grade fever, headache, or fatigue lasting a day or two. Clinical evidence supports that properly administered mesenchymal stem cell therapy is associated with transient mild reactions, not serious infections or deaths.
Rare, Serious Complications: Serious side effects are uncommon in proper medical settings and almost always result from protocol breakdowns. Infection is prevented through sterile technique; allergic reactions are rare with properly processed cells due to their immune-privileging properties; unwanted cell growth concerns primarily involve embryonic cells, not adult stem cells used in most clinics.
RMRM’s Systems-Based Optimization Approach
Your body is an integrated system, not isolated parts. Chronic inflammation, hormonal imbalances, and metabolic dysfunction create the environment where tissue degrades. Simply introducing cells without addressing the underlying biological terrain rarely produces lasting results.
At RMRM, we create the conditions where stem cells can survive, integrate, and function optimally. We offer a comprehensive toolkit of complementary therapies that work synergistically:
Hyperbaric Oxygen Therapy (HBOT): Delivers concentrated oxygen to support stem cell survival and integration.
Shockwave Therapy: Localizes cellular repair, provides immediate relief, and prepares tissue for regeneration.
Hormonal Optimization: Addresses endocrine deficiencies that impair repair.
Peptide Therapy: Supports healing, reduces inflammation, and optimizes metabolic function.
Advanced Diagnostic Imaging: We utilize MRI to ensure a full understanding of your health before recommendations.
Collaborative Rehab Network: We partner with select Boulder physical therapists for seamless pre- and post-procedure care.
This isn’t an assembly line. We personalize every protocol to your specific biology, goals, and preferences.
Read More: How Does Stem Cell Therapy Work
A Real-World Example: Beyond the Injection
Consider a patient who arrived with severe knee pain. A conventional clinic might have immediately injected stem cells. But we recognized something deeper.
During consultation, comprehensive testing revealed he was pre-diabetic and overweight. His weight was creating mechanical stress that would compromise any biological repair. Rather than simply injecting, we explained how to fully optimize his recovery:
- Immediate Relief: Shockwave therapy, 1-2 times weekly, provided significant pain relief
- Metabolic Intervention: We addressed insulin resistance with targeted medication and GLP-1 peptide support, connecting him with a local chef and nutritionist for meal preparation guidance
- Movement Re-education: Our Boulder physical therapy network taught him proper exercise patterns and is helping him maintain and exercise schedule
- Strategic Stem Cell Deployment: Only after 30 pounds of weight loss, biomarker improvement, and decreased systemic inflammation, did we harvest and deploy his autologous stem cells
- Post-Deployment Support: Hyperbaric oxygen therapy supported cell integration and function
Today, one year post-deployment, he is metabolically healthier, physically stronger, and preserved his native knee, avoiding surgical intervention entirely. This is the difference between transactional medicine and true biological infrastructure investment.
Read More: What Is Stem Cell Therapy And How Does It Work
Stem Cell Therapy as an Alternative to Surgery
Surgery represents a point of no return. Once hardware is implanted, it rarely comes out, and you’re exposed to potential complications from permanent implantation. Biological regeneration preserves your native anatomy while upgrading your body’s capacity to maintain it.
When surgeons are “on the fence” about necessity, the existence of biological alternatives becomes crucial. Yes, stem cell therapy requires out-of-pocket investment, but weighed against surgical costs, recovery time, lost productivity, and irreversible tissue alteration, biological optimization often proves the more prudent investment.
Choosing a Safe Provider
Your choice of clinic is the most important decision. Key questions:
Physician Credentials: Licensed medical doctor? Years of stem cell experience? Patient outcome data available?
Cell Sourcing & Processing: Where do cells come from? What screening occurs before use? Is the lab licensed (FDA certified)?
Systems Approach: Do they optimize the biological terrain, or just inject cells in isolation? Do they offer complementary therapies like HBOT, hormonal optimization, and rehab support?
Ongoing Care: Clear follow-up plan? How will they monitor progress? What happens if complications arise?
Read More: List Of Diseases Treated By Stem Cell Therapy
About Investigational Status
“Investigational” or “FDA-unapproved” does not automatically mean unsafe. Many effective therapy are investigational because they’re still being studied or developers haven’t pursued FDA approval. In Mexico, COFEPRIS-licensed clinics can offer investigational therapy under their regulatory framework: oversight through a different pathway, not no oversight.
In December 2024, the FDA approved Ryoncil (remestemcel-L-rknd), the first FDA-approved mesenchymal stem cell product in the U.S. for pediatric steroid-refractory acute graft-versus-host disease. This demonstrates that well-researched MSC therapies can successfully navigate full FDA approval.
How RMRM Prioritizes Safety
At RMRM, safety is the foundation of everything we do. Every stem cell therapy starts with a comprehensive evaluation. We assess whether therapy is appropriate for your specific situation and design personalized protocols based on your health profile. All procedures happen in licensed medical facilities with strict sterile protocols using ethically sourced, rigorously screened stem cells. We maintain structured follow-up monitoring and offer membership programs providing ongoing access to advanced diagnostics and personalized therapy planning.
Whether you desire comprehensive biomarker analysis and full-system optimization or prefer to proceed directly to autologous stem cell therapy, we personalize every aspect of your care.
What Comes Next
The most important step is connecting with a provider who prioritizes your safety and results equally. At RMRM, every patient deserves thorough evaluation, personalized therapy planning, and ongoing support.
Schedule a consultation to discuss whether stem cell therapy or other regenerative options make sense for your situation. Your health is too important for generic answers.
Disclaimer: This information is for educational purposes only and is not a substitute for professional medical advice. Stem cell therapies are investigational therapy that the FDA may not approve. Always consult with a qualified healthcare provider before beginning any therapy. Individual results vary, and no outcome is guaranteed.